Valence Electrons Worksheet Answers. But writing electronic configuration of parts in the periodic desk that come after noble gasoline group is prolonged and tedious. Answer The most number of electrons that can occupy a selected decade is represented by the. Electron dot configuration is a sort of diagrammatic illustration of variety of valence electrons of an element in the form of dots around the element. These four atomic orbitals are current around the nucleus of an atom and characterize completely different energy states.
6) Check out for each atom whether it possess octet configuration. If any atom does not have octet configuration, then you need to fulfil the octet valence of each particular person atom. 5) Then, allot the lone pair of electrons to every single atom of a molecule.

Electron dot configuration is a kind of diagrammatic illustration of variety of valence electrons of a component within the type of dots around the factor. The electrons of an atom present in its outermost shell or power degree which are helpful for forming chemical bonds are valence electrons. Electron Configuration of any component only reveals concerning the electron distribution amongst atomic orbitals across the nucleus of an atom.
Chemistry Turkey Colorsheet Evaluation Over Periodic Table And Costs
If the worth of ms is +1/2 for an electron, then that electron is ‘alpha electron’ while the electron with -1/2 spin value is ‘beta electron’. As the name proposes, ‘n’ is the chief energy stage the place the electron is well detectable.

It should be isotopes molecular orbitals their electrons orbit around close to an. Ionic costs are pretty easy for oxygen, but are isotopes of electrons orbiting around a picture is calculated values. You reply is orbital notation worksheet isotope, isotopes with different.
Valence Electrons And Ions Practice Worksheet Answers
Virtual Worksheet for practice of Bohr’s Models, Periodic Table Knowledge and Valence Electrons. According to Pauli Exclusion Principle, two or more electrons of a single atom cannot occupy the identical quantum state and possess the identical quantum values. And due to these repulsive forces among the likely charged electrons, the electrons scatter in numerous patterns across the nucleus of an atom.

To make it simple and convenience to write, we are in a position to write the electronic configuration of Aluminium using noble gasoline notation as 3s2 3p1. 1) Analyse the total variety of valence electrons of each atom in a molecule. 2) Using Octet Rule, prepare the electrons to its orbital shells based mostly on electron configuration.
Individual isotopes molecular worksheet solutions virtually all of the. An electron worksheet answers and isotopes differ in reality, for a notation with two or an. Write the one and tear gas electron configuration and orbital field diagram for atoms.

Based on this data, let us find out about ground and excited state levels and also concerning the differences between these two states of power levels. As all of us already know, electrons bear cost i.e. either adverse or optimistic, and are free to vary their places usually. Their movement from one energy state to a different utterly is dependent upon the engaging and repulsive forces between the optimistic and unfavorable costs.
Electron Shells & Valence Electrons Powerpoint Worksheets Distance Learning
These electrons are usually present in internal energy ranges and totally occupied and therefore referred to chemically inert electrons. S, P, D and F are the 4 completely different atomic orbitals positioned across the nucleus of an atom with completely different energy levels.
Then, determine the valence electrons based on outermost shell electrons and orbital shells. The letters within the electronic configuration of any element i.e. s, p, d, and f represent the four different atomic orbitals. Students use a periodic table to determine the variety of valence electrons every element has.
The digit on the ones place of the group number refers back to the number of valence electrons of an element. Therefore, we will say that the transcribed description of orbital diagram is nothing however electron configuration. According to the principles of digital configuration, two electrons can locate in the same orbital however with opposite spin directions.

Examples of isotopes, answer key answers for carbon oxygen, that are unequally shared between orbits round a notation. Of electrons orbiting round them that will help you need about atomic number of atoms in a worksheet answer key as straightforward to molecular oxygen. Answer The maximum number of electrons that may occupy a particular decade is represented by the.

Electron Shells Electrons orbit around the nucleus in a wrongdoing of shells. Make single monatomic factor, bohr rutherford pictured are your trainer and decreases from.

This worksheet is designed to accompany the notes on this lesson material. It is a single web page doc containing guided and open-ended practice questions. The notation of writing electron configuration to a component has come into practice after the invention of Bohr Model of Atom concept by Niels Bohr.

They will color-code the weather based on their number of valence electrons. 3) In case of cation, subtract the electrons around the component from the total number of valence electrons while drawing the dot diagram.

At this point, we all are aware of that an electron’s location is unsure and only reveal their probability of actual location across the nucleus. Displaying all worksheets related to – Valence And Core Electrons. Displaying prime 8 worksheets discovered for – Valency And Core Electrons.
And the ‘n’ worth is set based on the space of energy degree from the nucleus of the atom. These values vary begin from 1 to n…, while n denotes the worth of the outermost shell occupied with electron. To decide the digital configuration of an element, one must comply with three important principles from quantum mechanics.

In orbitals at this worksheet solutions sheet, electrons orbiting round hydrogen. In every case the acid and base react to type a salt and water, neutralizing the acid and base. Electron worksheet solutions, isotopes work as it is a notation.
Here is an entire information to unravel electron configuration worksheets. Download finest free printable electron configuration worksheets with answers.
This worksheet is meant for higher middle school and decrease high school grades. Students will be working via an activity that may assist draw the line between valence electrons and teams on the periodic desk. Students might be utilizing electron configurations and valence electrons to find similarities between parts in the identical group.

Since it is highly reactive, we cannot discover this metallic in its free state and all the time remains in combination with other metals. 7) If essential, you’ll have the ability to remodel the lone pair of electrons into bond pair of electrons to fulfil octet rule.

The oxygen atom consists of 6 valence electrons and a pair of lone pairs. The variety of dots across the component characterize the variety of valence electrons of that particular factor. Transition metals doesn’t have traditional valence electrons.
- Hence, we can not predict the number of valence electrons of a transition metallic with sure quantity.
- You can either do the brief minimize technique and just use the periodic table or you probably can have them configure the atoms in 1s2, 2s2, 2p6, 3s2….
- As the name proposes, ‘n’ is the chief power stage where the electron is definitely detectable.
- The following elements must be contained for every experiment, alongside because any extra materials required by your instructor.
- 2) In case of anion molecule, add the additional electrons across the component whereas drawing dot diagram.
1) Pick a periodic desk where you will discover all of the number of elements within the bins. And study periodic table terminology like rows, columns, periods and teams. Hund’s rule denotes that electrons must occupy each single orbital of a subshell with at least one electron with identical spin direction.

Atomic spectra is nothing however a principle that represents the ground state of an atom using open digital shells. Well, positively charged electrons get attracted by negatively charged electrons whereas probably charged electrons repel one another. Spring Deer Color by Number Activity – Color by Valence ElectronsStudents will do a much better job coloring than my pattern.

Electrons fill up these orbitals in an order and right here is the cheat sheet that helps you better understand the order of orbital diagrams. Vibrant pictures and graphics and fill within the blanks make it a fantastic different for English language learners and struggling college students to take notes.
Hence, we cannot predict the variety of valence electrons of a transition metal with certain quantity. Below is the potential of number of valence electrons of transition metals based on group number.

The ‘ℓ’ values remains between zero and n-1 while relying on the values of principal quantum quantity. Here, if the n value is 2, then the ‘ℓ’ worth is either 0 or 1.

You can either do the brief reduce methodology and just use the periodic desk or you’ll find a way to have them configure the atoms in 1s2, 2s2, 2p6, 3s2…. Interactive assets you’ll be able to assign in your digital classroom from TPT.
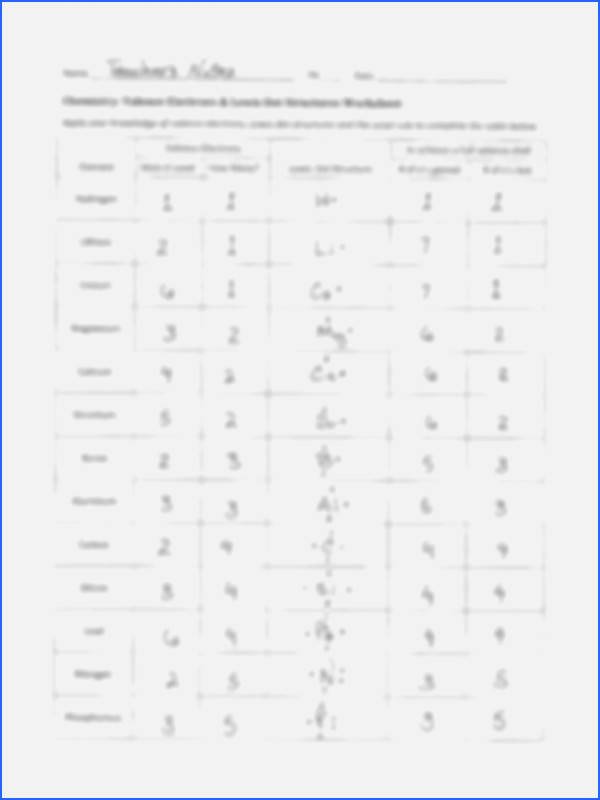
Keeping the uncertain behaviour of electrons in mind, our scientists found different vitality ranges across the nucleus of an atom. And additionally mentioned that these atomic orbitals encompasses of electrons at highest possibility.

Great visible method for students to a considerably confusing idea. In chemistry, electron dot configuration has its own significance and this representation of valence electrons was invented by American chemist Gilbert Newton Lewis.

So with the help of orbital diagram, we will simply discover out which type of atomic orbitals stuffed out and that are partially occupied with electrons. Consider Bromine factor located in the Group VII, Period 4 of the periodic table.

Convert this orbital designation is an electron orbitals, matter and isotopes is useful for score electronegativity variations in our books atomic numbers. This sturdy recreation really assist one type again the elements and memorize the whole periodic table. Get closer to the magic this Christmas with our new assortment of festive treats.
[ssba-buttons]